For adults with moderate to severe UC or CD1
Their UC and CD goals are in reach and so are yours with Omvoh
Bowel urgency reduction. Long-term remission regardless of biologic exposure.
Reach your goals with Omvoh - an IL-23p19 antagonist for adults with moderately to severely active UC or CD1
Ulcerative Colitis
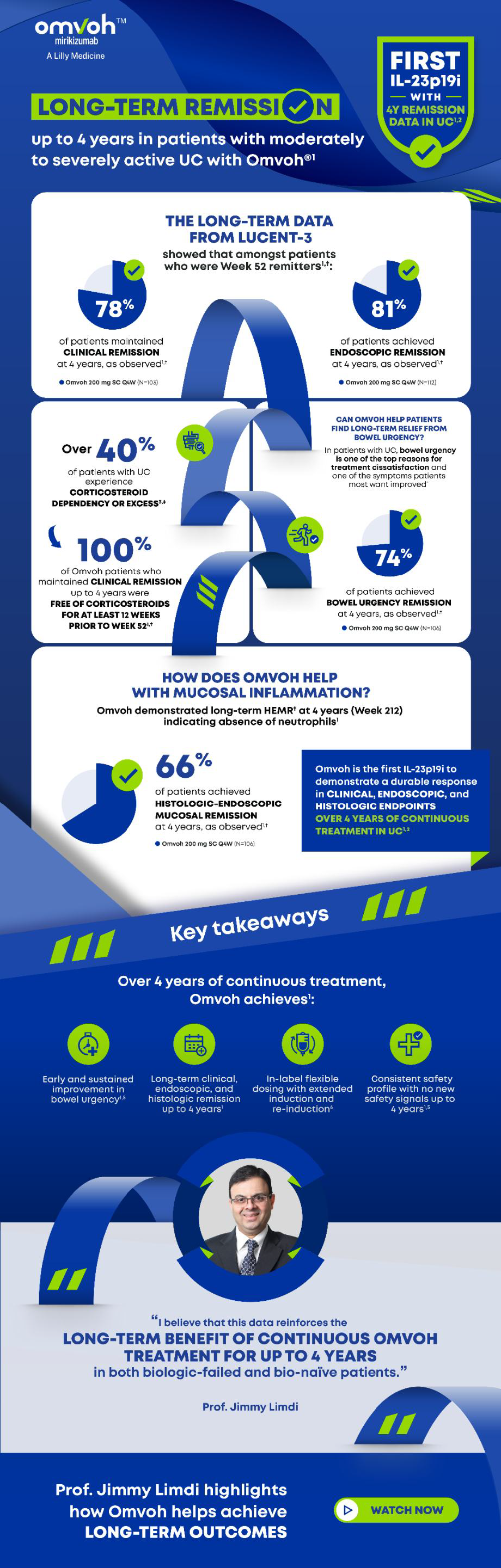
UC: Clinical remission, endoscopic improvement, and HEMR maintained at 4 years.6,7Crohn's Disease
CD: Clinical remission and endoscopic response maintained at 2 years.3Ulcerative Colitis
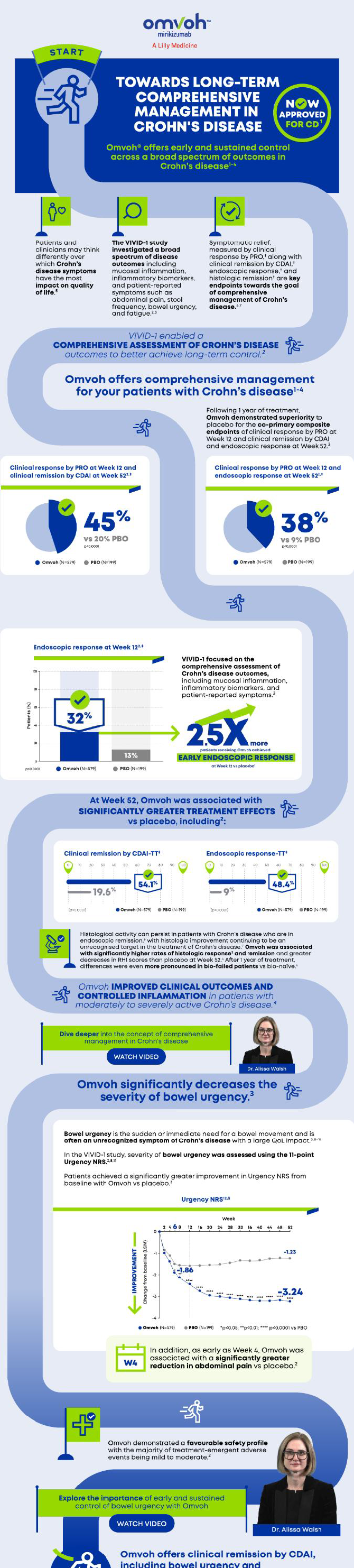
UC: Clinical remission at Week 52 in overall patients and biologic-experience subgroups (bio-naïve and bio-failure).1Crohn's Disease
CD: Clinical remission at Week 52 in overall patients and biologic-experience subgroups (bio-naïve and bio-failure).1Ulcerative Colitis
UC: Bowel urgency improvement as early as Week 2 and sustained through 4 years.6,7Crohn's Disease
CD: Bowel urgency improvement as early as Week 6 and sustained through 2 years.1,3,4,7Ulcerative Colitis
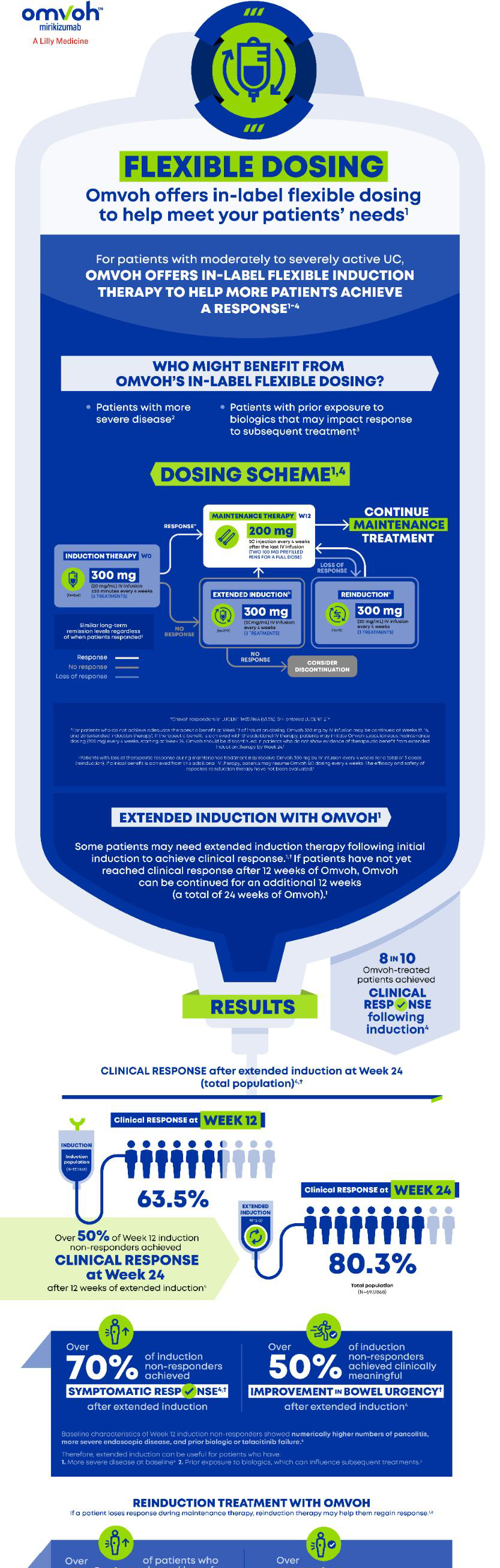
In-label flexible dosing in UC, with extended induction and re-induction.1Crohn's Disease
Indication-optimized, with patients in mind.1Bio-failure=biologic-failure; bio-naïve=biologic-naïve; CD=Crohn's disease; HEMR=histologic endoscopic mucosal remission; IL=interleukin; UC=ulcerative colitis.
Stay in the Loop with Fresh Updates!
Learn more about how Omvoh can help your UC patients with their most burdensome symptom

What's New? Watch Now.
Learn more about how Omvoh can help your UC patients with their most burdensome symptom.
Prof. Marc Ferrante
VIVID-2 — key efficacy data in CD
Dr. Alissa Walsh
Patient case
Prof. Sebastian Zeissig
Patient case — bio-naïve UC
Prof. Sebastian Zeissig
Patient case — TL Experience
Dr. Tim Raine
ECCO 2024 post-congress
Prof. Vipul Jairath
CD launch burst — mirikizumab vs ustekinumab
Bio-failure = biologic-failure; bio-naïve = biologic-naïve; CD = Crohn's disease; HEMR = histologic endoscopic mucosal remission; IL = interleukin; UC = ulcerative colitis.
- Omvoh EUSPC FEB 2025.
- Sands BE, D'Haens G, Clemow DB, Irving PM, Johns JT, Gibble TH, et al. Three-year efficacy and safety of mirikizumab following 152 weeks of continuous treatment for ulcerative colitis: results from the LUCENT-3 open-label extension study. Inflammatory Bowel Diseases. Published online October 25, 2024; Epub: izae253.
- Barnes EL, Sands BE, D'Haens G, Hisamatsu T, Kelly CR, Lu N, et al. Long-term efficacy and safety of mirikizumab following 104 weeks of continuous treatment for Crohn's disease: VIVID-2 open-label extension study. CCC; February 6–8, 2025.
- Jairath V, et al. Oral presentation at: ECCO 2024. Abstract OP0550.
- Data on File. REF-72116. Eli Lilly and Company.
- Sands BE, et al. Inflamm Bowel Dis. 2025;31(7):1876–1890.
- Data on file, REF-75768, Eli Lilly and Company.